 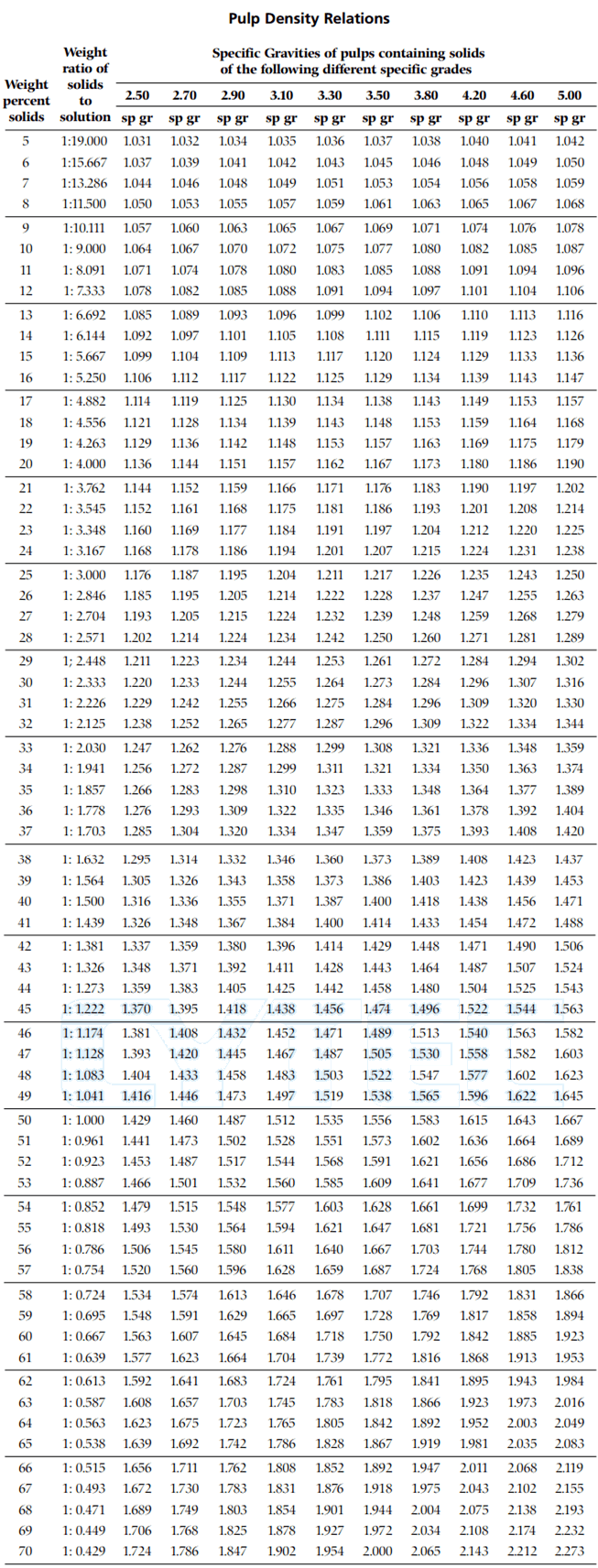
There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. Increases in temperature tend to decrease density since the volume will generally increase. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. This is a table of density (kg/L) and the corresponding concentration (Weight or Volume) of Ethanol (C 2 H 5 OH) in water at a temperature of 20☌. In the case of solids and liquids, the change in density is typically low. Density and Concentration Calculator for Mixtures of Ethanol and Water at 20☌. As one of the oldest commodities in the world, sugar is produced in over 120 countries and close to 80 of sugar comes from sugar cane, with sugar beets accounts for remainder. For human consumption, sucrose is extracted and refined from either sugarcane or sugar beet. 
It is produced naturally in plants and is the main constituent of white sugar.It has the molecular formula C 12 H 22 O 11. 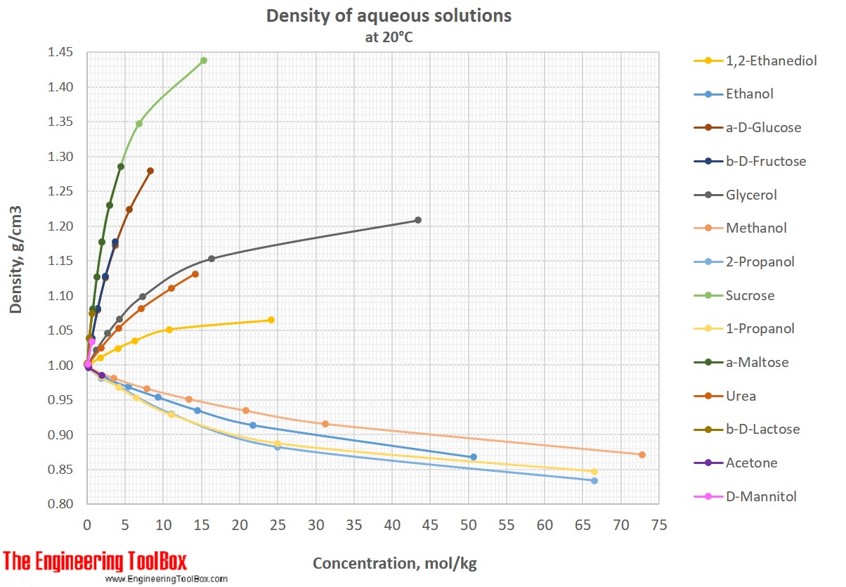
Note that density is also affected by pressure and temperature. Sucrose, a disaccharide, is a sugar composed of glucose and fructose subunits. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass Convert the density of water, 1 g/cm 3 to (a) lb/cm 3 and (b) lb/ft 3 and (c) g/cup. (This is the result of the tight interaction between the Sugar and Water molecules. As the Table Sugar becomes more concentrated, the density of the solution increases. There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. Consider solutions of Table Sugar in Water. However, it is important to pay special attention to the units used for density calculations. Calculate the density of (a) a piece of peach whose mass is 37.42 g and which, when submerged in water, increases the water level in a graduated cylinder by 35.80 ml (b) a 'heavy syrup' made by dissolving 4.00 cups (200 g/cup) of sugar in 5.0 cups (236.6 mL/cup) to make 7.00 cups of syrup. The calculation of density is quite straightforward. Sugar Water Solutions - Viscosities Temperature (o C) Absolute Viscosity (cP) Sucrose ( per weight) 20 40 60 0: 3.82: 14.8: 10: 2.66: 9.83: 113: 20: 1.97: 6.22: 56.7: 30: 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
